- FDA approved TECVAYLI (teclistamab-cqyv) plus DARZALEX FASPRO (daratumumab and hyaluronidase-fihj) for relapsed/refractory multiple myeloma.
- The treatment is for adults who have received at least one prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent.
- This approval introduces a potential new standard of care as early as the second line for 40% of patients experiencing disease relapse.
FDA Approval
The U.S. FDA has approved TECVAYLI (teclistamab-cqyv) combined with DARZALEX FASPRO (daratumumab and hyaluronidase-fihj) for treating adults with relapsed or refractory multiple myeloma (RRMM). This treatment is intended for patients who have undergone at least one prior therapy, including a proteasome inhibitor and an immunomodulatory agent.
New Treatment Approach
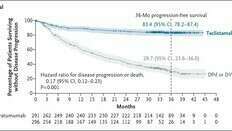
This approval offers a potential new standard of care as early as the second line, addressing the needs of the 40% of multiple myeloma patients who experience disease relapse. The combination works by priming and activating the immune system to target myeloma cells expressing the BCMA protein.
Expert Perspectives
Dr. Luciano J. Costa from the University of Alabama highlights the regimen's potential to redefine RRMM treatment by improving progression-free survival (PFS) and overall survival (OS) with a well-characterized safety profile. The option to use this regimen early is crucial due to the multiple relapses and reduced therapy responsiveness in RRMM patients.
Community-Based Treatment
Heather Ortner Cooper, President & CEO of the International Myeloma Foundation, emphasizes the importance of expanding community-based treatment options, allowing patients to receive care closer to home while respecting individual preferences. This approval enhances the therapeutic landscape, enabling oncologists to personalize treatment plans.

